- #CAPTURE ONE PRO 10 DOWNLOAD SOFTWARE#
- #CAPTURE ONE PRO 10 DOWNLOAD TRIAL#
- #CAPTURE ONE PRO 10 DOWNLOAD PROFESSIONAL#
With the rise of the internet in the mids, the obvious solution to some of these issues was the adoption of web-based software that could be accessed using existing computers at the investigational sites.ĮDC represents this new class of software. Usability and space constraints led to a lot of dissatisfaction among medical practitioners. The most significant shortcoming was that hardware e. Though effective, RDE brought with it several shortcomings as well. The system could then use a modem connection over an analog phone line to periodically transmit the data back to the sponsor, and to collect questions from the sponsor that the medical staff would need to answer. These early RDE systems used " thick client " software-software installed locally on a laptop computer's hardware-to collect the patient data. To address these and other concerns, RDE systems were invented so that physicians, nurses, and study coordinators could enter the data directly at the medical setting.īy moving data entry out of the sponsor site and into the clinic or other facility, a number of benefits could be derived. Historically, this information was collected on paper forms which were then sent to the research sponsor e. Nichol, Pickering, and Bollert offered "a controlled system for post-marketing surveillance PMS of newly approved NDA pharmaceutical products," with surveillance data being "entered into an electronic data base on site" at least as early as Clinical research data-patient data collected during the investigation of a new drug or medical device is collected by physicians, nurses, and research study coordinators in medical settings offices, hospitals, universities throughout the world. However, with the maturation of today's EDC solutions, much of the earlier burdens for study design and set-up have been alleviated through technologies that allow for point-and-click, and drag-and-drop design modules.ĮDC is often cited as having its origins in remote data entry RDE software, which surfaced in the life sciences market in the late s and early s. The net effect is to increase both the cost and risk to the study with insignificant benefits. This is often aggravated by two conditions.
#CAPTURE ONE PRO 10 DOWNLOAD TRIAL#
As a result, for an EDC to be economical the saving over the life of the trial must be greater than the set-up costs. EDC systems are used by life sciences organizations, broadly defined as the pharmaceutical, medical device and biotechnology industries in all aspects of clinical research, but are particularly beneficial for late-phase phase III-IV studies and pharmacovigilance and post-market safety surveillance.ĮDC can increase data accuracy and decrease the time to collect data for studies of drugs and medical devices. Ĭapture One Pro runs on the following operating systems: Windows.Ĭapture One Pro has not been rated by our users yet.An electronic data capture EDC system is a computerized system designed for the collection of clinical data in electronic format for use mainly in human clinical trials.ĮDC solutions are widely adopted by pharmaceutical companies and contract research organizations CRO. It was initially added to our database on.

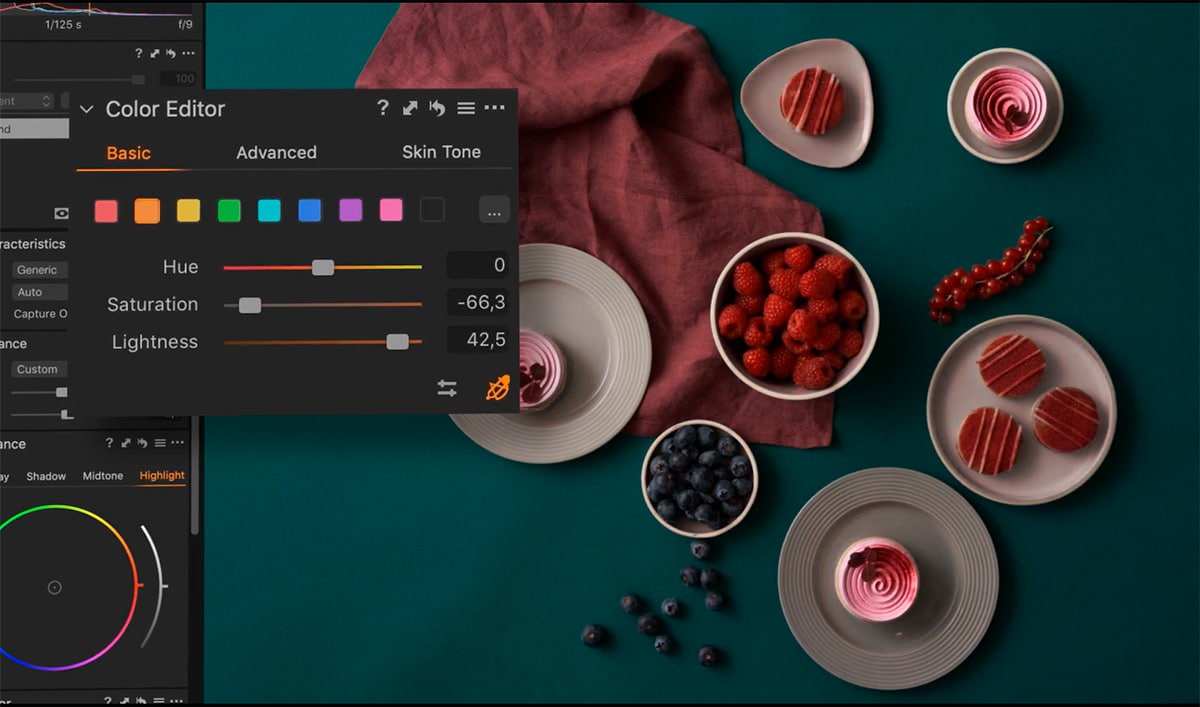
The latest version of Capture One Pro is 15.2.2, released on. It was checked for updates 31 times by the users of our client application UpdateStar during the last month. OverviewĬapture One Pro is a Demo software in the category Graphics Applications developed by Phase One. Capture One PRO has unlimited batch capability, multiple output files from each conversion, advanced color editor to create color profiles to suite your own color demands, IPTC/EXIF (meta data) support and much more. On top of this, Phase One's RAW workflow is renowned for its excellent image quality.

The main reason why Capture One PRO is the perfect choice for high volume photography is that it is designed to handle many images at a time.
#CAPTURE ONE PRO 10 DOWNLOAD PROFESSIONAL#
Capture One PRO is a RAW Workflow software designed for the professional digital photographer.
